Racetams are a class of potent synthetic brainpower-boosters that share a similar chemical structure.
- Neuroprotection. As they were originally intended, racetams help maintain brain health against degeneration, including age-related decline.
- Plasticity Support. Racetams are believed to promote the neuroregeneration and plasticity that is closely tied to learning, memory and long-range brain health.
- Neurotransmitter Balance. This class of nootropics has been suggested to help regulate acetylcholine and serotonin, brain chemicals tied to memory and mental performance.
- Brain Energy Metabolism. Racetams’ ability to boost cerebral circulation and nourish the brain with oxygen seems to augment its brain energy benefits.
Overview
Racetams are a group of nootropic drugs that share a similar chemical structure. Piracetam — the signature compound of the racetam family — was developed by Romanian psychologist and chemist Cornelius E. Giurgea in the 1960s. Piracetam was the first nootropic ever made, and indeed it was Giurgea himself who would go on to coin the term “nootropic” — meaning “acting on the mind” in Greek — in 1972.1
Racetams are mostly known for their metabolic and neurotrophic (neuron growth-promoting) effects. The other beneficial properties of these compounds include the stimulation of neurotransmission and brain-protective antioxidant activity.
Clinically, racetams are used to improve memory and overall mental function in patients suffering from dementia and related cognitive dysfunctions. Despite this, racetams are far more widely used by healthy individuals as a cognition-enhancing drug, and are a popular topic of discussion among “biohackers” seeking to improve mental capacity.
 The majority of racetam research has been performed in countries of the former Soviet Union, where racetams are still available by prescription for improving cerebral circulation and facilitating recovery after traumatic brain injury.2 3
The majority of racetam research has been performed in countries of the former Soviet Union, where racetams are still available by prescription for improving cerebral circulation and facilitating recovery after traumatic brain injury.2 3Main Types of Racetams
Piracetam
Piracetam (2-pyrrolidone acetamide, 2-Oxo-1-pyrrolidine, Memotopril, Fezam (with cinnarizine), UCB6215), a derivative of the neurotransmitter γ-aminobutyric acid (GABA), is the original and most studied racetam.4
Piracetam has demonstrated positive effects on memory, learning, depression, anxiety, and overall cognitive function.56Although it is commonly used in the treatment of dementia and brain injury, piracetam is far more popular as a mental performance booster and study aid in younger populations.
The mechanism by which piracetam exherts its effects remains unclear, although researchers theorize that it may work by restoring brain cell membrane fluidity.

Aniracetam
Aniracetam (1-p-anisoyl-2-pyrrilidinone, Ro 13-5057, CAS 72432-10-1, 1(4-methoxybenzoyl)-2-pyrrolidinone, Ampamet, Memodrin, Pergamid) is a fat-soluble, fast-acting racetam. Similar to piracetam, it improves memory and cognitive function affected by dementia, but has a stronger relaxing, anxiolytic, and mood-enhancing effect.
Moreover, Aniracetam indirectly contributes to neurotransmission via activating nicotinic acetylcholine receptors in the brain, and has been suggested to improve verbal fluency and social behavior.8
Aniracetam’s main limitation is that it has a shorter half-life than piracetam and therefore wears off faster. Furthermore, the evidence for its effects is largely restricted to rat and mice models.9 10 11
Oxiracetam
Oxiracetam (4-hydroxy-2-oxo-pyrrolidinoacetamide, ISF-2522) is structurally related to piracetam and commonly referred to as a “speedy” racetam. 12
Oxiracetam has been shown to improve memory and learning in both young and old laboratory animals more than piracetam.13 14The main mechanism of its action appears to be an increase of the neurotransmitter acetylcholine at the neuron synapse level.15
However, oxiracetam can have undesirable consequences as well; it’s fast-acting stimulatory effects can lead to extreme tiredness and a “crash” several hours after ingestion. This may be one of the reasons why, similar to aniracetam, it is no longer used in a clinical setting.
Pramiracetam
A piracetam derivative, Pramiracetam (CI-879, Diisoprop-yl-(2-oxopyrrolidin-1-yl)acetamide, CI879) is one of the most effective nootropics used for the purpose of cognitive enhancement.
Preliminary data generated in experiments on young and old rats suggests that pramiracetam could be used to facilitate long-term memory formation.16
Although its mechanisms are not well understood, the pharmacological effect of Pramiracetam might be mediated by an increase in high-affinity choline uptake. Pramiracetam might also enhance acetylcholine synthesis and improve memory and cognitive deficits associated with sustained traumatic brain injury.17
Noopept
While piracetam seems to improve only early stages of the memory recall process, noopept stimulates the memory consolidation and retrieval steps as well.
However, the neuroprotective effects of this compound are yet to be confirmed, and there is currently no evidence of cognition-enhancing activity. In addition, there is not enough data regarding the safety of noopept in humans.
Phenylpiracetam
Phenylpiracetam (Phenotropil, Carphedon, (RS)-2-(2-oxo-4-phenylpyrrolidin-1-yl)acetamide) is a phenylated analog of piracetam developed in the USSR in 1983.19
Research on rodents has shown that phenylpiracetam may have memory-preserving and enhancing effects, in addition to acting as an antidepressant, anxiolytic and anticonvulsant.
Phenylpiracetam is believed to be more potent than piracetam and is used for the enhancement of memory, mental concentration, and learning capacity. Interestingly enough, phenylpiracetam is also able to reduce the negative effects of sleep deprivation on higher brain functions.
Nefiracetam
Nefiracetam (DM-9384, N-(2,6-dimethylphenyl)-2-(2-oxopyrrolidine-1-yl)-acetamide) is another cognitive enhancer derived from piracetam, although structurally and functionally it is more similar to aniracetam, another fat-soluble racetam. Both of these compounds are known for their anti-amnesic effects, and are used in cognitive decline therapy.
Nefiracetam has been shown to have antidepressant and anticonvulsant effects in rodents, and to lessen apathy in depressed stroke patients. However, unlike most racetams, nefiracetam has no acute effects and has to be supplemented for a prolonged period of time; as such, it might be beneficial for the preservation of long-term memory processes.20 21 22
The physiological actions of nefiracetam appear to be mediated by the stimulation of GABAergic and cholinergic signaling pathways of the brain.23 24
As demonstrated by preliminary studies in dogs, nefiracetam’s major downside is that it may be toxic.25
Are Racetams Legal?

However, you can still purchase bulk racetam powder from some US suppliers, and it’s also possible to purchase racetams from other countries, such as Canada.
Meanwhile, in most countries of the European and former Soviet Union racetam drugs are available by prescription, where they are used for helping with cognitive deficiency. More on Nootropic Legality
Racetams’ Potential Nootropic Bio-Activities
The common underlying mechanism of action of racetams is not fully understood. The results of preclinical studies suggest that these substances may work by:
Improving energy balance in brain cells
First off, racetams may improve the function of the mitochondria — the energy-generating cell structures — in neurons. In particular, activation of the adenylate cyclase enzyme by piracetam may increase synthesis of cAMP, providing higher turnover of ATP, the body’s main energy exchange molecule. In turn, these processes lead to the intensification of cellular respiration and glucose utilization.26
Enhancing synaptic plasticity
In addition, racetams seem to influence synaptic plasticity — the ability of neuron synapses to strengthen or weaken over time — by modulating the function of excitatory neurotransmitter receptors. In particular, racetams have been shown to slow desensitization and abolish deactivation of α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors for the neurotransmitter glutamate. These receptors mediate fast synaptic transmission in the central nervous system (CNS).27
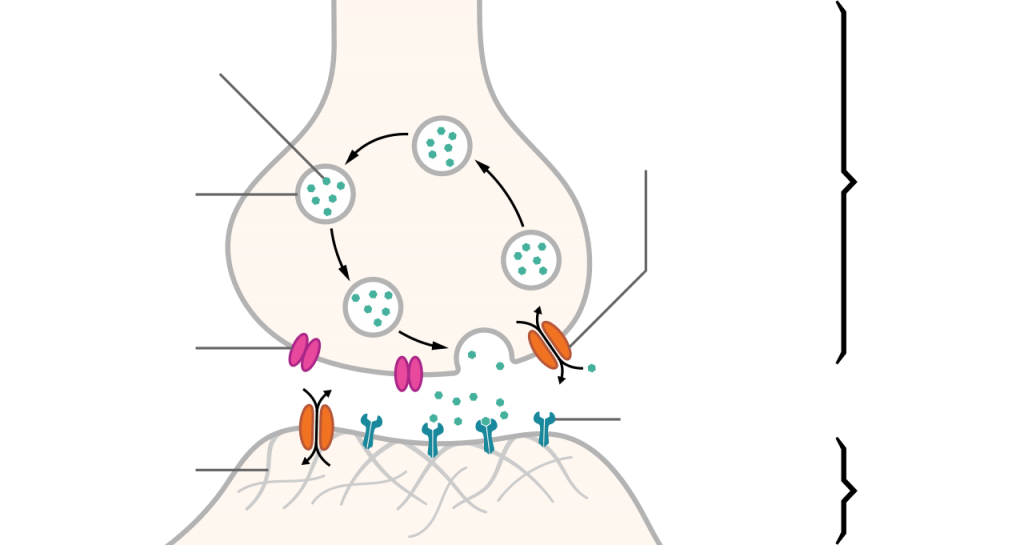
Synapse between two neurons. By Thomas Splettstoesser [CC BY-SA-4.0], via Wikimedia Commons
Activating anabolic processes in CNS
Racetams may also increase the synthesis of RNA, proteins, and phospholipids, which can improve the metabolism of phosphatidylcholine and phospho-ethanol-amine — the main components of brain cell membranes. 28
Increasing the levels of neuromodulators
Furthermore, racetams could possibly stimulate neuromodulation — a process through which the activity of multiple neurons is modified — by supporting the synthesis and utilization of the neurotransmitters acetylcholine and serotonin. This could, in turn, improve communication between the brain’s left and right hemispheres.
Improving cerebral circulation
Moreover, racetams appear to improve the resistance of neurons to hypoxia (lack of oxygen) and toxins, and might also enhance cerebral blood circulation and oxygen flow.29 30 31
Stabilizing membranes
Lastly, racetams may increase the fluidity of brain cell membranes. In particular, piracetam has been shown to enhance the hydrocarbon core fluidity of hippocampal membranes isolated from Alzheimer’s patients and healthy elderly individuals in a concentration-dependant manner.32
Racetam Research
Although human trials support the use of racetams for brain disorders such as dementia, there is currently no definitive evidence backing the purported cognition-enhancing effects of racetams in healthy people. Moreover, current research is largely restricted to animal data, and what little human research exists is limited by small sample sizes and lack of long-term outcomes.
Piracetam
Piracetam 400 mg/day may improve verbal learning in healthy individuals
This early double-blind study tested the effect of 400 mg piracetam daily on memory in 16 healthy college students. The subjects had to learn series of words presented as stimuli. Although no effects on memory were detected after 7 days of treatment, verbal learning was significantly improved after 14 days.
- The researchers concluded that “human brain power can be selectively increased” with piracetam supplementation33
Piracetam 8 g/day appears to help improve memory function in Alzheimer’s
This double-blind study tested the effectiveness of piracetam in helping with Alzheimer’s. Thirty-three early Alzheimer’s patients took piracetam (8 g) or placebo daily for a year. Although there was no substantial improvement in either group, piracetam resulted in significantly better memory test results.
- The reseachers concluded that “our results support the hypothesis that long-term administration of high doses of piracetam might slow the progression of cognitive deterioration in patients with AD“34
Aniracetam
The aim of this study was to determine the efficacy of aniracetam alone or in combination with cholinesterase inhibitors (ChEIs) in dementia therapy. A total of 276 patients (mean age 71) diagnosed with cognitive disorders of variable severity were examined at baseline and at 3, 6, and 12 months of treatment.
Aniracetam significantly improved the emotional state of the patients after 3 months. Moreover, aniracetam-treated patients with mild dementia demonstrated improved functionality after 3 months and better cognitive performance after 6 months when compared to the ChEIs group. Aniracetam alone was also found to be more effective than the combination treatment.
- The researchers concluded that “Aniracetam is a promising option for patients with cognitive deficit of mild severity”35
Oxiracetam
In this double-blind trial, 65 patients (mean age 71) with dementia were given oxiracetam (800 mg) or placebo twice daily for 12 weeks. The oxiracetam-treated group showed significant improvement on the quality of life scale, as confirmed by a number of neuropsychological tests. The researchers noted the necessity of long-term studies for a thorough evaluation of the therapeutic effects of Oxiracetam.
- The researchers concluded that “Oxiracetam may be used in the pharmacological treatment of Alzheimer’s disease or multi-infarct dementia”36
Dosage Notes
- Piracetam: 480 mg – 1200 mg
- Aniracetam: 400 mg -1500 mg
- Noopept: 10 mg – 30 mg
- Nefiracetam: 150 mg -900 mg
- Oxiracetam: 1200 mg -2400 mg
- Phenylpiracetam: start with 100 mg daily as its long-term usage is associated with tolerance
Side Effects
Overall, racetams are well-tolerated, and are rarely associated with serious side effects. Infrequent adverse reactions associated with Racetams include headaches, insomnia, agitation, tremors and intense sweating, dry mouth, and sometimes skin rashes.
However, it’s important to note that while some researchers have suggested racetams to be non-toxic, not enough research has been performed to confirm this.
Supplements in Review Recommendation
- Piracetam Pure Powder, 800 mg.
Although research is lacking, the popularity of racetams speaks for itself. Racetam research is limited and inconclusive, and there’s no evidence that they work in healthy individuals. Despite this, off-label racetam use remains popular and many people report significant improvements in various aspects of cognition, such as memory and concentration.
Low doses of piracetam powder is a cheap and safe way to see if racetams work for you. Considering that the effects of racetams seem to vary between individuals, it’s best to start out with a low dose like 800 mg to see if it feels right. If you notice improvements in cognition, you can try try upping your Piracetam dose. Or, you can try stacking it with a powerful (but lightly researched) racetam like Noopept to further raise the racetam brain-boosting activity.
References
Leave a Reply